
Modern scientists have found smaller particles that make up the protons, neutrons, and electrons, although the atom remains the smallest unit of matter that can't be divided using chemical means. For atoms with a high number of electrons, relativistic effects come into play, since the particles are moving at a fraction of the speed of light. Rather than the circular orbits of Rutherford's model, modern atomic theory describes orbitals that may be spherical, dumbbell-shaped, etc. The electron can potentially be found anywhere in the atom but is found with the greatest probability in an atomic orbital or energy level. Quantum mechanics led to an atomic theory in which atoms consist of smaller particles. This, in turn, led to Werner Heisenberg's uncertainty principle (1927), which states that it's not possible to simultaneously know both the position and momentum of an electron. Louis de Broglie proposed a wavelike behavior of moving particles, which Erwin Schrödinger described using Schrödinger's equation (1926). In 1913, Frederick Soddy described isotopes, which were forms of an atom of one element that contained different numbers of neutrons. Several discoveries expanded the understanding of atoms. But Rutherford was an industrious researcher who many remarkable contributions to science, including three discoveries that revolutionised our view of matter. The model and its validation in 1908 by Jean Perrin supported atomic theory and particle theory.īohr's model explained the spectral lines of hydrogen but didn't extend to the behavior of atoms with multiple electrons. This year is the 100 year anniversary since Ernest Rutherford published his seminal paper describing his discovery of the atomic nucleus. In 1905, Albert Einstein postulated that Brownian motion was due to the movement of water molecules. 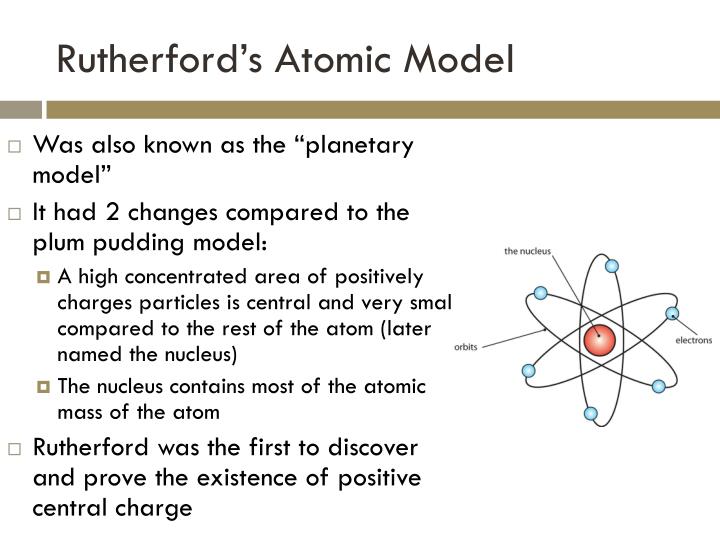
Avogadro's law made it possible to accurately estimate the atomic masses of elements and made a clear distinction between atoms and molecules.Īnother significant contribution to atomic theory was made in 1827 by botanist Robert Brown, who noticed that dust particles floating in water seemed to move randomly for no known reason. In 1811, Amedeo Avogadro corrected a problem with Dalton's theory when he proposed that equal volumes of gases at equal temperature and pressure contain the same number of particles. His oral presentation (1803) and publication (1805) marked the beginning of the scientific atomic theory. He proposed that each chemical element consists of a single type of atom that could not be destroyed by any chemical means. .png)
Dalton's law of multiple proportions drew from experimental data. These theories didn't reference atoms, yet John Dalton built upon them to develop the law of multiple proportions, which states that the ratios of masses of elements in a compound are small whole numbers. Ten years later, Joseph Louis Proust proposed the law of definite proportions, which states that the masses of elements in a compound always occur in the same proportion. In 1789, Antoine Lavoisier formulated the law of conservation of mass, which states that the mass of the products of a reaction is the same as the mass of the reactants. He also has element 104 named after him, dubbed Rutherfordium.It took until the end of the 18th century for science to provide concrete evidence of the existence of atoms. He was also knighted within his lifetime. 
Rutherford's interesting facts are that he had predicted the existence of a neutron before it was ever officially discovered. This equation led him to see that hydrogen produced this equation, and that it must have it's own particle making up the nucleus, and he dubbed it the proton. As for the proton, he had discovered that through usage of mathematics, creating the nuclear reaction equation. This experiment made it clear that there was a core stopping the particle from going through, and he dubbed it the nucleus. He had discovered the nucleus through his famous Gold Foil Experiment.In this experiment, he shot alpha particles out to a sheet of golf foilf, but not all of them made it through, with some even bouncing back. This discovery majorly changed the way atoms were viewed and modeled, as now they had to factor in the nucleus and the protons. Rutherford's biggest contribution to the atomic theory was the discovery of both the nucleus of the atom, and of protons in the atom. Thomson's replacement at Cambridge, after the retirement of Thomson. He eventually went through college and was able to go overseas to wok with J. He had read a science book at the age of 10, and that's when his love for science first took shape, as he was so interested he performed experiments from what the book had told him. He was talented in both intellect and in sports themselves, a heavenly combo for any scientists. Ernest Rutherford was born in Brightwater, New Zealand, and lived from 1871-1937.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
